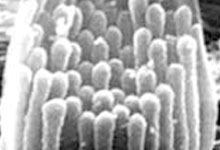
Since its inception, the NOHR Foundation has provided over $9.8 million for 510 separate research grants. In 2012, we are funding 10 "Seed Money" projects. Ultimately, discoveries about auditory function will lead to strategies to treat, prevent or cure hearing loss, deafness and related disorders.
ONE-YEAR SEED MONEY GRANTS
The Seed Money Program is the NOHR Foundation's primary focus, making funds available for scientists to pursue new ideas and directions in their research.
Seed Money grants awarded by the NOHR Foundation for 2012 are supporting projects by the following principal investigators:
- Justin Aronoff, Ph.D., is an Advanced Research Associate in the Communication and Neuroscience Division of House Research Institute in Los Angeles
Dr. Aronoff�s project is related to bilateral cochlear implantation. It investigates different implant designs with the goal of improving wearers� understanding of speech in difficult listening situations, such as noise. Although two implants electrically process sounds received in both ears independently, the sounds may interfere with each other during the brain�s neural-encoding process, affecting perception. Experiments will test different locations of electrodes and different allocations of frequency information among the electrodes of two implants. How these manipulations enhance wearers� ability to understand speech in noise will be measured.
https://www-scf.usc.edu/~aronoff/ - Martin L. Basch, Ph.D., is a Postdoctoral Associate in the Department of Neuroscience at the Baylor College of Medicine (Houston, TX)
Dr. Basch is studying the embryonic development of a cochlear structure called the stria vascularis. This tissue generates special fluid that is rich in potassium and is essential for normal hearing. Strial defects are implicated in presbycusis (age-related hearing loss) and many human syndromes involving profound hearing loss. The goal is to identify genes that could be manipulated to repair or restore damage to or degeneration of the stria vascularis resulting from heredity or aging.
https://neuro.bcm.edu/groveslab/ - Christopher Clinard, Ph.D., is an Assistant Professor in the Department of Communication Sciences and Disorders at James Madison University
Dr. Clinard is investigating a specific neural activity in the brain, called phase locking, which is involved in the perception of frequency in sounds, and is related to speech-understanding. Older adults, even those with normal hearing sensitivity, may have difficulty comprehending speech. Dr. Clinard�s experiments will characterize how aging affects the ability to perceive small frequency differences in sounds. The work is related to aural rehabilitation of adults with hearing loss.
https://www.csd.jmu.edu/auditoryperceptionlab.html - Soroush Sadeghi Ghandehari, Ph.D., is a Postdoctoral Fellow in the Department of Otolaryngology at Johns Hopkins University School of Medicine
Dr. Ghandehari is investigating the relationship between two types of sensory cells, afferents and efferents, of the vestibular (balance) system. Afferent cells called calyces enclose vestibular �hair� cells and convey information to the brain about rapid head movements. Efferent cells provide �feedback� from the brain and strongly affect calyces. Dr. Ghandehari is studying mechanisms in cell synapses, (connections where signals are transmitted), after damage, as from surgery for tumors called schwannomas. The work is related to designing vestibular rehabilitation programs and treatments utilizing drugs.
https://neuroscience.jhu.edu/ElisabethGlowatzki.php - Maria Eugenia Gomez-Casati, Ph.D., is an Assistant Researcher at the Instituto de Investigaciones en Ingenieria Genetica y Biologia Molecular in Buenos Aires, Argentina
Dr. Casati�s research involves the relationship between the afferent and efferent neural systems of the cochlea. Afferent fibers send sound information from the cochlear �hair cells� to the brain; efferent fibers that project from the brain provide inhibitory �feedback� that affects ability to hear. The work is related to the relationship between early hearing loss caused by noise damage to the ear�s sound system and maturation of the brain�s auditory area. Information about how noise exposure in daily life affects age-related hearing loss will help in development of compounds to prevent noise-induced trauma. - Michelle Hastings, Ph.D., is an Assistant Professor in the Department of Cell Biology and Anatomy at the Chicago (IL) Medical School, Rosalind Franklin University
Dr. Hastings� project involves development of a potential genetic treatment for hearing loss from degeneration of spiral ganglion neurons after loss of cochlear �hair cells.� Spiral ganglion neurons are critical for the preservation of hearing. She will test synthesized molecular compounds designed to alter a protein gene-splicing defect in ways beneficial to spiral ganglion neurons. The goal is development of a method for systematically delivering therapeutic molecules to the ear to prevent spiral ganglion neuron loss after �hair� cell loss. - Ambrose Kidd, Ph.D., is a Research Scientist at Washington University in St. Louis, MO
Dr. Kidd�s project is investigating a potential drug therapy to prevent or mitigate presbycusis, or age-related hearing loss. This common disorder afflicts about one half the population over 75 years of age. He is studying whether the combination of an anti-oxidant drug and an anti-epileptic drug, which separately delay age-related deafness loss in mouse models, together reduce hearing loss at lower dosages. Side effects of dosages effective at preventing hearing loss will also be measured.
https://otocore.wustl.edu - Rutherford M. Ongkeko, M.D., Ph.D., is an Associate Project Scientist at the University of California, San Diego
Dr. Ongkeko�s project seeks to understand how a specific genetic defect affects the hereditary disease neurofibromatosis 2. Afflicted individuals develop tumors called vestibular schwannomas, which can cause deafness, imbalance, brainstem compression and death. His project studies whether the genetic mutation leads to loss of tumor-suppressing capabilities by affecting stem cell proliferation and renewal. - Bradley J. Walters, Ph.D., is a Postdoctoral Research Fellow in the Department of Development Neurobiology at St. Jude Research Hospital in Memphis, Tennessee
Dr. Walters� project investigates inner ear hair cell regeneration in a new animal model, the opossum, a marsupial. Hair cell regeneration has been extensively studied in lower vertebrates, such as birds and fish, which are able to regenerate damaged or lost �hair cells.� It has been speculated that the evolutionary position of the opossum affords it similar capability. In addition, marsupials, while not placental mammals like humans, possess a coiled cochlea like humans. It is hypothesized that if their �hair� cells possess regenerative capacity, it would indicate that the mammalian cochlea could possibly possess this capability also. - Yasheng Yuan, M.D., Ph.D., is a Postdoctoral Research Fellow in the Department of Otolaryngology, Eaton-Peabody Laboratory, Massachusetts Eye and Ear Infirmary, Harvard Medical School
The goal of Dr. Yuan�s research is to develop a therapy to replace lost or damaged auditory cells of the cochlea, called spiral ganglion neurons, through transplantation of embryonic stem cells. This project aims to test hearing function of transplanted cells, including threshold and frequency perception. It also aims to correlate functional measurements with calculations of numbers and sites of new spiral ganglion neuron formation in the cochlea.
The NOHR Foundation provided one investigation 200,000 for two years (2008-2009) for an inner ear hair cell regeneration program; provided a total of 2.5 million to two investigations for five years (2001-2005) for hair cell regeneration programs; and provided one program a total of $300,000 for a three-year (2001-2003) inner ear hair cell regeneration project
ACCOMPLISHMENTS:
TWO-YEAR PROGRAM (2008-2009)
Tatjana Piotrowski, Ph.D. & Alejandro Sanchez Alvarado, Ph.D., University of Utah School of Medicine
In a bioinformatics and genetic investigation in zebrafish,
- a mutant in which hair cells form normally but fail to properly regenerate once damaged (as occurs in mammals) has been identified
- genes in a cell communication pathway (Wnt) that respond to the injury and death of hair cells have been discovered and cloned
https://research.stowers.org/piotrowskilab/Grants.php
FIVE-YEAR PROGRAM (2001-2005)
Neil Segil, Ph.D. and Andrew Groves, Ph.D. House Ear Institute, Los Angeles
Studying embryonic and postnatal mice, they have discovered:
- auditory supporting cells from newborns can spontaneously change into hair cells under culture conditions
- a gene called p27Kip1 blocks this process in the inner ear
- in mutant mice lacking p27Kip1, cells were able to divide and make hair cells
https://www.hei.org
Michael Lovett, Ph.D. and Mark Warchol, Ph.D. ,Washington University, St. Louis
In microarray studies of cochlear and vestibular avian ear genes,
- specific genes that are switched on and off during the process of inner ear hair cell regeneration after damage have been discovered
- new genomic techniques to shut down/off genes and investigate subsequent changes in other genes and in the regeneration process were developed
- genetic components of regeneration in balance structures have been revealed
THREE-YEAR PROGRAM (2001-2003)
Douglas A. Cotanche, Ph.D. Children's Hospital, Boston
- developed embryonic mammalian cochlear cultures to serve as neural stem cell transplant recipients
- discovered neural stem cells transplanted into cochlear cultures do differentiate into cochlear cells
Johns Hopkins University
 |
National Institute on Deafness and Other Communication Disorders www.nidcd.nih.gov |
 |
Association for Research in Otolaryngology www.aro.org |
 |
American Academy of Otolaryngology www.entnet.org |
 |
Write My Essay For Me by Academized.com academized.com |